Production of the 4 basic GANSes: ZnO, CO2, CuO, CH3
This article is part of the KF Plasma Times January 2019
Keywords: zinc oxide, carbon dioxide, copper oxide, hydrocarbon, gans production.
Published: January 2019.
Subject
This paper provides the reader with an easy to follow, step-by-step guide on how to produce the four basic GANSes, which are in various ways connected to our bodies. Other resources relating to this topic exist on the KF Wiki [1] and the KF SSI YouTube channel [2]. The basic theory behind the production procedure is explained together with the affinity of each of these GANSes to the particular aspects of the human (or any other animal’s) body.
Taking a step back, let’s first consider what GANS actually is. GANS is the abbreviation for Gas in Nano-state of Solid and as explained by M.T. Keshe in his second book, "This condition of GANS of matter comes about at ambient temperature and pressure when due to internal Gravitational and Magnetic fields’ strength of the atom the atom’s physical appearance changes and the atomic structure of the gas changes to a compact configuration of the solid. The atom of the same gas becomes and behaves like a solid but with totally new properties and characteristics which were never known when the atom was in its other three initial known states (gas, liquid and solid)." (Keshe, 2011).
Method
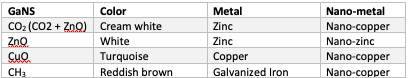
There are many methods through which one can produce GANSes. Traditionally, this is achieved through the use of two (or more) metal plates or coils, one of which is Nano-coated. Table 1 gives an overview of which metals are used in the production of the four GANSes. Other methods exist, for example, GANS of vitamins, food and other substances can be prepared through the use of caustic soda or CO2 GaNS and plasma water. The process in a GaNS production box is similar to the process of absorption of food or air – no matter crosses the wall of the lungs or intestine, but a certain condition is created to attract the field of the elements of Oxygen (lungs) and other matter consumed (intestine). In this way human body (and the GANS production box) creates a condition of a ‘magnet’, which attracts fields of a specific strength (being the weaker and hence dictating the direction of the flow). The attracted fields, in the salty water environment, slow down and transform into the matter state of a given element.
The complete list of materials required for the production of any of these four GANSes is as follows:
- Plastic box (optionally Nano-coated for faster production)
- One metal plate or coil
- One Nano-coated metal plate or coil
- Pure sea salt and distilled water
- Copper wire for short-circuiting of the metals
- Green LED
- Two alligator clips (optional if plates are not drilled)
- Nano-coated plastic spoon to harvest amino-acids
- 2 x Plastic or glass containers to store amino-acids and GANS
- Syringe to collect the GANS or to remove excess water
Production procedure
Step 1)
Depending on which GANS one wants to produce (as per Table 1), Nano-coat one of the plates/coils following any of the available video or text guides [3]. Prior to this process, if using metal plates instead of coils, drill a hole in a corner of both plates (this eliminates the need for the use of alligator clips). If no drill available, alligator clips can be used.
Step 2)
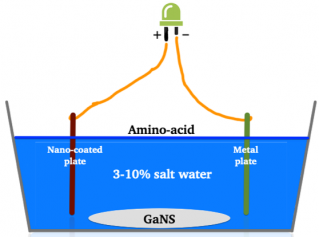
Hang the two plates in the production box, opposite each other at a distance of several centimeters - as per Fig. 1. Plates or coils should not touch the bottom of the container but instead should be fixed 1-2’’ above its bottom. If they sit on the bottom of the box, this will slow down or prevent GANS from being produced.
Step 3)
Connect the two plates via a copper wire either by going through the drilled holes or using alligator clips. Optionally when connecting the plates, one can place a green LED in this circuit. The use of an LED light in the circuit helps to create the flow of energy between the plates. As with every GANS production, connect the anode (longer piece of wire = positive pole) of the LED with the copper wire that leads to the Nano-coated metal plate/coil, and the cathode (negative pole) with the copper wire that leads to the metal plate/coil (Fig. 1).
Step 4)
Fill up the GANS production box with a 3.5-10% sea salt, distilled water solution (make sure the salt is not contaminated by any minerals). Place the box away from other boxes and reactors and leave alone for couple of weeks. The time needed to produce each of these GANSes depends on a number of factors, such as type of GANS produced, materials available in the proximity of the production box, water salinity, distance between the plates, quality of the Nano-coating, purity of metals and other.
Step 5)
When one observes the deposit of the GANS at the bottom of the box, or the plates become too salty or worn out, it is time to harvest the GANS. Set the plates aside and with a Nano-coated plastic spoon, collect the amino acid which floats on the water’s surface. Store it in a clean glass or plastic container and top it up with distilled water not to dry out.
Step 6)
Either use a syringe to collect the GANS sediment from the bottom of the production box or carefully remove the excess water with it and pour the sediment into a separate glass or plastic container to store it. Washing of the GANS to reduce salinity is optional and depends on the application area. For more information and pictures, detailed guide on the production of the four basic GANSes is available on the KF wiki [4].
Useful Tips
- Make sure you wash your Nano-plate of any caustic soda residue, before using it in the GANS production.
- Handle Nano-materials with care. Do not scratch it, be gentle with it and never stack it up.
- Use caustic soda Nano-coating method which results in more permanent Nano-layers than fire-coating.
- Make sure the salt you are using has no impurities.
- You can produce GANSes not only through the use of sea or table salt (NaCl) but also using other salts such as Magnesium or Potassium.
- Nano-coat your GANS production box for better results or simply reuse one from a previous GANS production of the same GANS as during the production process walls of the container get Nano-coated.
- As water from your GANS production box evaporates and decomposes into GANS, some of the salt crystals can deposit at the bottom, which reduces water salinity. Top up your boxes with distilled water and if needed add some salt into the distilled water used for topping up.
- Keep you GANS production boxes at least 3-5 meters apart in order not to influence the process in each box and to achieve the expected results.
- Use plates/coils which are not too thin as they can break either during the Nano-coating or GANS production processes.
- Every GANS produced is different in strength – think of it as isotopes of the same element. Therefore, in order to cover a wide spectrum of fields, add your e.g. freshly produced CO2 GANS into the jar/container with previously produced CO2 GANS. Over time, your CO2 GANS has many different ‘flavors’ to it and can be more effective in any application area.
How does it work
Let’s consider the CO2 GANS production in which a Zinc and a Nano-coated copper plates are used. Atomic weight of Copper is just over 63.5 (69% of the copper isotopes is Cu63 and almost 31% is Cu65) and Zinc is 65 (just over 49% is Zn64, other isotopes are in the range of 66-68) [5]. In the Nano-coating process, any metal loses around 5% of its atomic weight. Therefore, Copper reduces to around 59 atomic weight. The difference between the two plates (65 and 59) is 6 and it is the atomic number (number of protons in the nucleus) of Carbon. Therefore, the CO2 production box creates a plasma field between the two plates of the strength 6, which like a magnet, attracts Carbon present in the atmosphere around the box. In plasma, stronger feeds the weaker to find balance, and hence the field created in the box is the weaker part. In the salty water solution, Carbon oxidizes in the presence of the plasma of Oxygen and deposits as a CO2 GANS at the bottom. The side effect of the CO2 GANS production is the presence of ZnO GANS. The Nano-coated copper plate creates a condition for attraction of Zinc from the Zinc plate, which in turn oxidizes to become the ZnO. As a result, the CO2 box produces a mixture of CO2 and ZnO GANS, where the ratio of the two depends on the setup. More details can be found on the KF wiki page on the CO2 GANS production [6].
GANS of CH3 is produced in a similar way and is a 2-stage process. Firstly, the Nano-copper (59) interacts to find balance with the layer of Zinc of the galvanized Iron. This ‘magnet’ for Carbon attracts this element and as a consequence the CO2 GANS gets produced and deposits at the bottom of the box. Secondly, the fields of the Nano-coated copper plate interact with Iron (atomic weight close to 56, with almost 92% of Fe56) and create a ‘magnet’ for H3. Tritium attracted from the atmosphere binds with Carbon fields already present in the salty water environment and deposits as the CH3 GANS at the bottom of the production box.
CuO and ZnO GANS productions use the same metals to produce it. For CuO it is Copper and Nano-copper, for ZnO it is Zinc and Nano-zinc. The Nano-coated metal plate attracts/pulls elements out from the other plate, which then oxidize in the plasmatic water environment and deposit at the bottom of the container in the form of GANS. GANSes acquire the affinity towards the environment which produced it. Therefore, the CO2 GANS has affinity towards Copper and Zinc, as well as towards Sodium (as NaCl is present in the water). Moreover, whatever materials are used in the Nano-coating process, the produced GANS also has affinity towards them e.g. using Magnesium Hydroxide Mg(OH)2 in the Nano-coating process. Therefore, each of the produced GANSes differs in strength as it is affined to the elements and environment which produced it. In other words, these are the common denominators Knowledge Seekers hear so much about in various context.
Observations
During the GANS production one observes the following. Firstly, oxygen bubbles appear on the Nano-metal plate/coil. After couple of days (or weeks, depending on the setup) one can notice a sediment deposition at the bottom of the production box and a thin layer of amino-acid building on the water’s surface. Often, salt crystals build up and deposit on the part of the Nano-plate which is above the water’s surface.
Usage
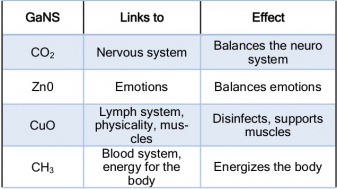
The GANS and amino-acid production is the way life started on this planet. It all started in the salty water environment of the Oceans and surrounding matter and GANS state. During GANS production process, the two metal plates in a salty water environment create a condition for attraction of certain elements e.g. Carbon, Zinc, Copper, Oxygen, or Hydrogen. These are the basic elements, which life on this planet is based on. Table 2 shows the connection of these four basic GANSes to the human being. GANSes are the basis of the Plasma Technology and are applicable in many areas, such as Food and Agriculture, Health, Energy, Decontamination, Space Travel and other. Amino-acids collected from different GANS production boxes are of different strength in respect to each other, as they get attracted by different strength `plasmatic magnets` created between two metal plates. GANS is also used to prepare the liquid plasma by mixing it with distilled water and collecting the water above the sediment. For applications in Health and Agriculture, amino-acid is added in the process of preparing the liquid plasma to strengthen its connection to living organisms, which are all based on amino-acids.
Acknowledgements
The content presented in this publication is based on the knowledge and discoveries of M.T. Keshe and other Knowledge Seekers who over the course of the years documented it in the form of videos and wiki articles.
Supplementary material
An extensive guide on how to produce these four basic GANSes is available on the KF Wiki. These guides contain links to YouTube videos and other handy resources. Please visit https://en.kfwiki.org/wiki/Category:Nano-Coating_and_the_Production_of_GANS for more details.
References
- https://kfwiki.org/
- https://www.youtube.com/channel/UCtQzN7XLiLvTpyUnQAa1mRw
- https://en.kfwiki.org/wiki/Category:Nano-Coating_and_the_Production_of_GANS#Nano-Coating_Using_NaOH
- https://en.kfwiki.org/wiki/Category:Nano-Coating_and_the_Production_of_GANS#Different_Types_of_GANSes
- https://ptable.com/
- https://en.kfwiki.org/wiki/CO2_GANS
- https://store.keshefoundation.org/store/product/GANS_Reserve_Bottle/
- Keshe, M. (2011). The Structure Of The Light. Stichting the Keshe Foundation.